NeuroBo Pharmaceuticals' Novel GLP1R and GCGR Dual Agonist, DA-1726, Shown to Elicit Superior Weight Loss Efficacy Compared to Semaglutide and Tirzepatide in Preclinical Models
Preclinical Data Also Show DA-1726 Effectively Reduces Body Weight and Glycemic Control
Data Presented in One ePoster Theater Discussion and Two General Poster Presentations at the
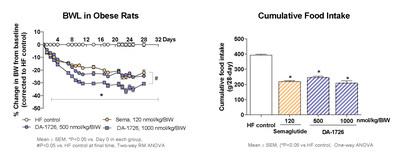
"DA-1726, a long acting OXM analog that binds and activates both GLP-1 and glucagon receptors using a well understood mechanism, has shown weight loss efficacy, despite similar or higher food intake, in a dose-dependent manner, that is better than SEMA in diet-induced obese (DIO) rats (32.6% for DA-1726 at a high dose compared to 24.0% for SEMA, p<0.05)," stated
"In preclinical mice models, DA-1726 also showed the ability to improve plasma glucose insulin and Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) as compared to Cotadutide (COTA), another OXM analog. Importantly, DA-1726 showed superior plasma triglyceride (TG) reduction and a similar reduction of total cholesterol (T-CHO) compared to COTA. In a comparative preclinical mouse study with GLP1R/glucose-dependent insulinotropic polypeptide receptor (GIPR) dual agonist, Tirzepatide (TIR), DA-1726 showed similar efficacy on weight loss, despite consuming more food. However, DA-1726 was more efficacious in improving plasma metabolic parameters such as glucose, TG, and T-CHO compared to TIR, indicating differential metabolic effects caused by GCGR agonism. Taken together, these data suggest that DA-1726 is a well-balanced GLP-1 receptor and glucagon receptor dual agonist and we anticipate that DA-1726 will have effective weight loss and glycemic control in humans," concluded
"DA-1726 is a long-acting, novel peptide drug candidate in preclinical development with therapeutic promise for obesity and NASH, in preparation for a phase 1 clinical trial for obesity," stated
Abstract Title: A Novel GLP1R/GCGR Dual Agonist, DA-1726 Elicits Weight Loss Superior to Semaglutide in Diet-Induced Obese Rats
Poster: 1676
Authors:
Presenter:
Session:
Date and Time:
Location: Poster Halls B-C
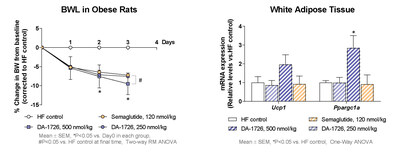
DA-1726 showed superior weight loss efficacy compared to Semaglutide (SEMA) in diet-induced obese (DIO) mice. In this study, a dose-response of DA-1726 was evaluated to determine the maximum efficacy in DIO rats which are known for translatability to human studies. The half-life of DA-1726 was longer in rats compared to mice, however the in vitro activity of DA-1726 against rat glucagon receptor was less potent than in mice, and plasma protein binding was approximately 10-fold higher in rats. Therefore, the effective dose for rats was set higher than for mice. DA-1726 was injected twice a week for approximately 4 weeks, and a dose-dependent and significant weight loss effect was observed. The minimum effective dose in DIO rats was found to be 250 nmol/kg. Then, the maximum body weight loss effects of DA-1726 (500 and 1000 nmole/kg) and SEMA were compared in the DIO rat model. DA-1726 showed an excellent weight loss effect compared to SEMA (32.6% for DA-1726 at a high dose vs. 24.0% for SEMA, p<0.05). The low-dose DA-1726 group showed a similar effect compared to SEMA in spite of consuming more food. The high-dose DA-1726 group showed similar food intake as SEMA, however it led to a higher weight loss effect. After repeated dosing, the high-dose DA-1726 increased the expression of energy metabolism-related genes (Ucp1, Ppargc1a) in white adipose tissues (WAT), supporting increased energy expenditure. In order to confirm whether this increased gene change was a direct effect leading to weight loss or an indirect effect caused by weight loss, changes in gene expression were assessed after single dosing. DA-1726 (250 and 500 nmol/kg) induced small but significant weight loss 3 days after a single injection (-7.6% and -9.5% vs. control). However, the genes expression (Ucp1, Ppargc1a) in WAT was increased only in the 500 nmol/kg-treated group suggesting a direct effect. Therefore, DA-1726 is expected to elicit significant weight loss effects in humans with a new mechanism of action.
DA-1726 showed equal or greater efficacy with higher food intake compared to Semaglutide in reducing body weight
DA-1726 showed significant increase in white adipose tissue gene expression suggesting energy expenditure's direct impact on weight loss
Abstract Title: Differentiated Metabolic Effects of DA-1726, a Balanced GLP1R/GCGR Dual Agonist
Authors:
Poster: 1668
Session: ePoster Theater
Date and Time:
Location: Exhibit Hall (ePoster Theater A)
Session: General Poster Session
Date and Time:
Location: Poster Halls A-B
The pharmacological effect of DA-1726 was evaluated compared to other competitor peptides. DA-1726 showed superior efficacy compared to another GLP1R/GCGR dual agonist, Cotadutide (COTA), in reducing body weight (14.4% for DA-1726 and 6.4% for COTA vs. diet-induced obese (DIO) mice control at 30 nmol/kg, p<0.05) and improving plasma glucose, insulin, and Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) (-91% for DA-1726 vs. -52% for COTA) in DIO mice. In addition, DA-1726 showed superior plasma triglyceride (TG) reduction and similar reduction of total cholesterol (T-CHO) compared to COTA. The data suggests that the balanced dual agonist DA-1726 was superior to a GLP1R-biased dual agonist in metabolic effects. Additionally, enhanced glucagon action of GLP1R/GCGR dual agonists may increase the risk of hyperglycemia when GLP1R agonism is reduced. DA-1726 was injected for 3 weeks and a glucose tolerance test was performed at 72 hours after the last dosing. DA-1726 did not impair glucose tolerance even at minimal plasma concentrations during repeated dosing up to 100 nmol/kg. In the case of repeated dosing of 100 nmol/kg, the plasma drug concentration at 72 hours after the final injection is similar to the concentration that induces about 50% activation of receptors under 4% human albumin condition. In a comparative study with a GLP1R/glucose-dependent insulinotropic polypeptide receptor (GIPR) dual agonist, Tirzepatide (TIR), DA-1726 showed a similar maximum efficacy on weight loss (31.2% for DA-1726 and 31.3% for TIR vs. DIO control, p<0.05). However, DA-1726 was more efficacious in improving plasma metabolic parameters such as glucose, TG, and T-CHO compared to TIR, indicating differential metabolic effects caused by GCGR agonism. Taken together, this data suggests that DA-1726 is a well-balanced GLP1R/GCGR dual agonist that effectively reduces body weight and glycemic control.
DA-1726 showed similar efficacy with higher food intake compared to Tirzepatide in reducing body weight
Copies of the presentation materials are available on the
About
For more information, please visit www.neurobopharma.com.
Forward Looking Statements
Certain statements in this release may be considered forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, including without limitation, statements about the closing of the offering of securities. Forward-looking statements are predictions, projections and other statements about future events that are based on current expectations and assumptions and, as a result, are subject to risks and uncertainties. Many factors could cause actual future events to differ materially from the forward-looking statements in this release, including, without limitation, those risks associated with our ability to execute on our commercial strategy; the timeline for regulatory submissions; the ability to achieve results in human clinical trials that are similar to those received in preclinical animal trials; NeuroBo's ability to achieve similar results in future preclinical trials; regulatory steps and potential regulatory approval of NeuroBo's current and future product candidates, the ability to realize the benefits of the license agreement with
Contact:
+1-917-633-6086
mmiller@rxir.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/neurobo-pharmaceuticals-novel-glp1r-and-gcgr-dual-agonist-da-1726-shown-to-elicit-superior-weight-loss-efficacy-compared-to-semaglutide-and-tirzepatide-in-preclinical-models-301863706.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/neurobo-pharmaceuticals-novel-glp1r-and-gcgr-dual-agonist-da-1726-shown-to-elicit-superior-weight-loss-efficacy-compared-to-semaglutide-and-tirzepatide-in-preclinical-models-301863706.html
SOURCE